|
Photochemically intercalated sodium ion enhances the photoelectrocatalytic activity of WO 3. Sodium containing mixed oxides are promising catalysts and photocatalysts. Sodium soaps have a higher melting temperature (and seem "harder") than potassium soaps. Most soaps are sodium salts of fatty acids. Two equivalent images of the chemical structure of sodium stearate, a typical soap. In compounds, sodium is usually ionically bonded to water and anions and is viewed as a hard Lewis acid. The most important sodium compounds are table salt (Na Cl), soda ash (Na 2 CO 3), baking soda (Na HCO 3), caustic soda (NaOH), sodium nitrate (Na NO 3), di- and tri- sodium phosphates, sodium thiosulfate (Na 2 S 2O 3♵H 2O), and borax (Na 2 B 4O 7♱0H 2O). Sodium compounds are of immense commercial importance, being particularly central to industries producing glass, paper, soap, and textiles. This framework disintegrates when dissolved in water and reassembles when the water evaporates. A) Selenium is one of the known elements.See also: Category:Sodium compounds The structure of sodium chloride, showing octahedral coordination around Na + and Cl − centres.A) Aluminum oxide is a single, chemically pure compound.Hence a compact disc is not chemically pure.ī) The regions of different composition indicate that a compact disc is a heterogeneous mixture. A) A compact disc is a solid material that contains more than one element, with regions of different compositions visible along its edge.A) Orange juice contains particles of solid (pulp) as well as liquid it is not chemically pure.ī) Because its composition is not uniform throughout, orange juice is a heterogeneous mixture.It is usually separated from tea leaves by filtration.ī) Because the composition of the solution is uniform throughout, it is a homogeneous mixture. A) Tea is a solution of compounds in water, so it is not chemically pure.If its composition is uniform throughout, it is a homogeneous mixture. If a substance is not chemically pure, it is either a heterogeneous mixture or a homogeneous mixture.If a substance can be separated into its elements, it is a compound. If it is pure, the substance is either an element or a compound. Decide whether a substance is chemically pure.aluminum oxide, a white powder that contains a 2:3 ratio of aluminum and oxygen atoms.Identify each substance as a compound, an element, a heterogeneous mixture, or a homogeneous mixture (solution). Any given spoonful of soup will contain varying amounts of the different vegetables and other components of the soup. Vegetable soup is a heterogeneous mixture. All solutions would be considered homogeneous because the dissolved material is present in the same amount throughout the solution.Ī heterogeneous mixture is a mixture in which the composition is not uniform throughout the mixture. The amount of salt in the salt water can vary from one sample to another. The difference is that the composition of the substance is always the same. Often it is easy to confuse a homogeneous mixture with a pure substance because they are both uniform. The salt water described above is homogeneous because the dissolved salt is evenly distributed throughout the entire salt water sample. It retains its composition and properties.Ī homogeneous mixture is a mixture in which the composition is uniform throughout the mixture. Only the form of the salt is changed when it is dissolved into water. A mixture is a physical blend of two or more components, each of which retains its own identity and properties in the mixture. You may dissolve a small amount of salt or a large amount into a given amount of water. /GettyImages-sb10067655by-001-1e3eb6ce621f4747b863b631b5cad181.jpg)
Salt easily dissolves in water, but salt water cannot be classified as a pure substance because its composition can vary. All samples of sodium chloride are chemically identical. It is considered a pure substance because it has a uniform and definite composition. Ordinary table salt is called sodium chloride. 
A material composed of two or more substances is a mixture. 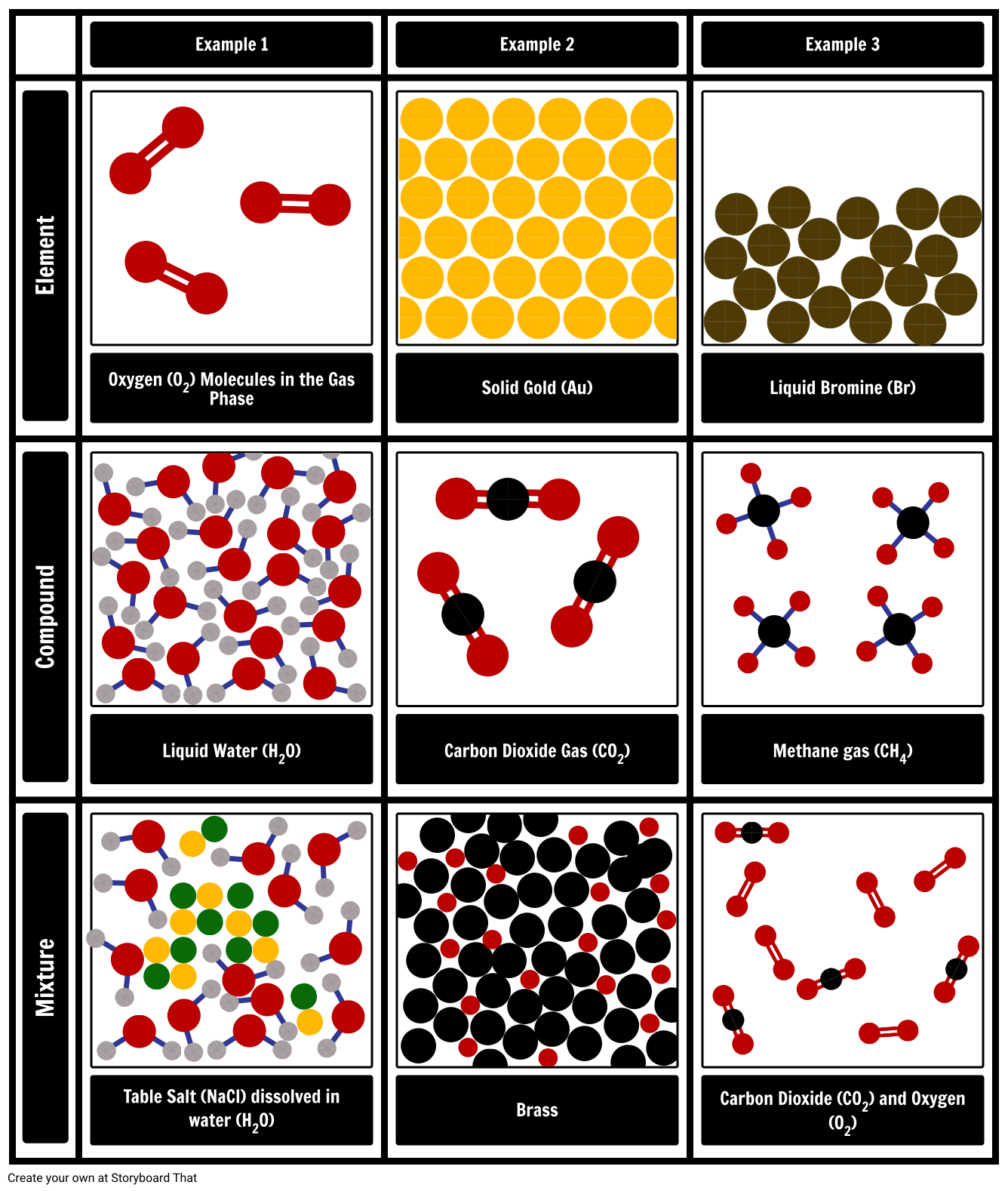
A pure substance is a form of matter that has a constant composition (meaning it's the same everywhere) and properties that are constant throughout the sample (meaning there is only one set of properties such as melting point, color, boiling point, etc. Matter can be classified into two broad categories: pure substances and mixtures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
